A Look Inside: Batteries from Usage to Reuse
In this activity, you will explore what batteries are made of; how they are used; what happens when they stop working or are damaged, defective, or recalled (DDR); and how to dispose of them properly through recycling.
Facilitation Tips and Potential Student Answers
Be sure to scroll to the bottom of the Educator Overview until you see STUDENT CONTENT BELOW THIS LINE. Interleaved in the Student Content section, you will see educator facilitation tips and potential student answers displayed in red, italic text when applicable.
Activity Title:
A Look Inside: Batteries from Usage to Reuse
Description:
In this activity, students will explore what batteries are made of; how they are used; what happens when they stop working or are damaged, defective, or recalled (DDR); and how to dispose of them properly through recycling. Students will develop an understanding of battery materials and the recycling process through exploring different battery types, watching videos of recycling facilities, and sharing their learning through creative projects.
Target Grade Level:
Grades 5-8
Discipline or Course (Audience):
STEM
Time Frame:
Two to three 45 - 60 minute sessions
Suggested Grouping:
Pairs or groups of 3-4 students
Key Vocabulary:
Recycling facilities, contamination
Educator Prep:
- When planning to show a video on YouTube, if you do not have access to ad-free video playing, you might consider putting the video link into a Google Slide deck and showing from there, rather than showing directly from YouTube.com. Sometimes this process can reduce or remove ads that might otherwise be inserted into the video.
Materials:
- Variety of battery types and sizes, whether physical objects or images. Try to include alkaline and lithium batteries; AA, AAA, C, D; cylindrical, rectangular, button.
- Images of devices that use batteries
- Chart paper or digital tools, smart board projector, etc.
- Digital or paper journal to record student thinking
- Graphic Organizer: Types of Batteries Supporting Material - Battery Types - Graphic Organizer
- Internet access
- YouTube Video: Web Link - How Millions Of Batteries Are Recycled Every Day | YouTube
- Web Link - Battery Types | Cirba Solutions
- YouTube Video: Web Link - The Ways and Whys of Recycling with Jeff Snyder of Rumpke Waste & Recycling
- YouTube Video: Recycling Lithium Batteries in 15 Minutes. How do they do that? (Teacher Use)
KEY SAFETY NOTES and Background Information:
- Batteries SHOULD NEVER go into curbside trash or recycling bins.
- Batteries can be dangerous if damaged. If contents leak out, this can cause a chemical contamination risk. Lithium batteries that are damaged, defective, or recalled (DDR) can pose increased risk of going into thermal runaway, which can cause a fire.
- Look for signs of damage like bulging, cracking, bloating, leaking, burn marks, etc. Such indications show that could be increased risk of a thermal event (fire) or contamination. DDR batteries should be safely and properly disposed of as quickly as possible.
- Alkaline batteries can be recycled and processed into materials that can be used as micronutrients in fertilizers.
- End-of-life lithium batteries are recycled and processed into "black mass" containing metals like lithium, nickel, cobalt, etc., that can be used to make new batteries.
- Lithium primary (metal) batteries are single-use batteries. A primary battery is a fuel cell where the fuel is held in or on the electrodes instead of in a tank. The electrodes therefore are being consumed in the production of electrical energy.
- Lithium-ion batteries are rechargeable and use lithium compounds rather than pure lithium metal as the electrode material. Lithium-ion batteries are constructed with a layered structure, consisting of alternating layers of anode, cathode, and the separator/electrolyte.
Background Information Links:
- Lithium vs Lithium-Ion Battery Guide - Cell Saviors
- Lithium Battery and Lithium-Ion Battery - BEASFA
- Lithium-Ion vs Lithium Batteries: Key Differences - CheckFire
- Is a Li-Ion Battery a Lithium Battery? - The Battery Tips
- Lithium vs Lithium-Ion Batteries - RD Batteries
Optional Experience:
Consider a real-world connection experience. Visit a local recycling facility that accepts end-of-life batteries (not all do!), or invite a guest speaker from your local municipality's waste management, household hazardous waste department, or school district's facilities to provide students with insights about the battery recycling process.
Teacher Directions:
Begin the lesson through engagement. Display pictures of a battery (or bring real examples) and ask the following question:
Where does this battery go when it stops working?
Engage in a conversation that prompts students to think about the batteries they encounter in their everyday lives. Record answers either on chart paper or digitally.
Activity Preparation Directions:
- Gather battery types and pictures.
- Print Supporting Material - Battery Types - Graphic Organizer
- Record size, labels, possible materials, and where these batteries can be used.
- Safety considerations/disposal of lab materials: Recycle batteries properly.
Mini-Lab Observe and Classify:
Pass out a variety of batteries and put students into pairs or groups. You can include real batteries and images of batteries. Ask students to observe and classify the batteries by the following attributes:
- Size
- Labels
- Uses
- Possible materials
Have them discuss and justify their choices. For example, these are Duracell AA batteries and can be used in toys, remotes, etc.
Special Population Accommodations:
Recommendations for supporting English Language Learner (ELL) students:
- Follow district guidelines for supporting ELL students.
- Consider sensory supports such as physical movement or podcasts.
- Use graphic organizers, anchor charts, and visuals to support and scaffold.
- Offer sentence frames and/or academic discourse stems for tasks such as explaining reasoning or comparing and contrasting ideas.
- Pre-teach vocabulary within the content.
- Pair with a peer buddy and assign a bilingual or high-proficiency English-speaking peer for support.
- Create opportunities for multiple ways to demonstrate understanding, such as through visual, oral, or written means.
- Model thinking out loud to showcase reasoning and problem-solving skills.
Recommendations for supporting Gifted & Talented (GT) students:
- Follow district guidelines for supporting GT students.
- Gifted students thrive when given choices and opportunities to explore the complexities of the content.
- Encourage students to explore across disciplines.
- Allow for opportunities for self-directed inquiry and research.
- Encourage reflection on learning strategies and thinking processes used.
- Provide executive functioning support and check-ins as needed.
Recommendations for supporting students with Individualized Education Programs (IEPs) and 504s:
- Follow the required accommodations based on the student's IEP or 504 plan.
- Provide small-group and individualized instruction as needed.
- Reduce cognitive load through chunking, scaffolding, and visual supports.
- Offer feedback frequently.
- Allow for extended time.
- Allow for breaks as needed.
- Provide modeled examples and clear single-step instructions prior to students engaging in independent or group work.
- Review evidence-based interventions providing support in small groups or individualized support as needed.
Remote Learning Adaptations:
This activity is appropriate for remote learning; no adaptations or modifications are necessary.
STUDENT CONTENT BELOW THIS LINE
We take our garbage out to the dumpster or pick-up site every time the trash can gets full, but what should we do with batteries that can no longer be used? Do you ever wonder…
Essential QuestionWhere does a battery go when it stops working? |
Materials:
- Variety of battery types and sizes (alkaline, lithium, etc.)
- Images of devices that use batteries
- Chart paper or digital tools, smart board projector, etc.
- Digital or paper journal to record thinking
- Learning Tool: Supporting Material - Battery Types - Graphic Organizer
- Internet access
- YouTube Video: Web Link - How Millions Of Batteries Are Recycled Every Day | YouTube
- Weblink: Web Link - Battery Types | Cirba Solutions
- YouTube Video: Web Link - The Ways and Whys of Recycling with Jeff Snyder of Rumpke Waste & Recycling
Safety Notes:
- When using technology, engage in safe, legal, and ethical behavior; this applies to devices, software, and online interactions.
- Take care when handling batteries, especially lithium-ion and damaged, defective, or recalled (DDR) batteries. Follow teacher directions.
KEY SAFETY NOTES and Background Information:
- Batteries SHOULD NEVER go into curbside trash or recycling bins.
- Damaged batteries pose fire risks or chemical contamination risks if they leak.
- Lithium-ion batteries (DDR) can go into thermal runaway, causing a fire.
- Look for damage: bulging, cracking, bloating, or leaking. DDR batteries must be disposed of properly and quickly.
- Alkaline batteries are processed into micronutrients for fertilizers.
- End-of-life lithium batteries are processed into "black mass" (containing lithium, nickel, cobalt) to make new batteries.
- Lithium primary (metal) batteries are single-use batteries. A primary battery is a fuel cell where the fuel is held in or on the electrodes instead of in a tank. The electrodes, therefore, are being consumed in the production of electrical energy.
- Lithium-ion batteries are rechargeable and use lithium compounds rather than pure lithium metal as the electrode material. Lithium-ion batteries are constructed with a layered structure, consisting of alternating layers of anode, cathode, and the separator/electrolyte.
Batteries Have a Hidden Life
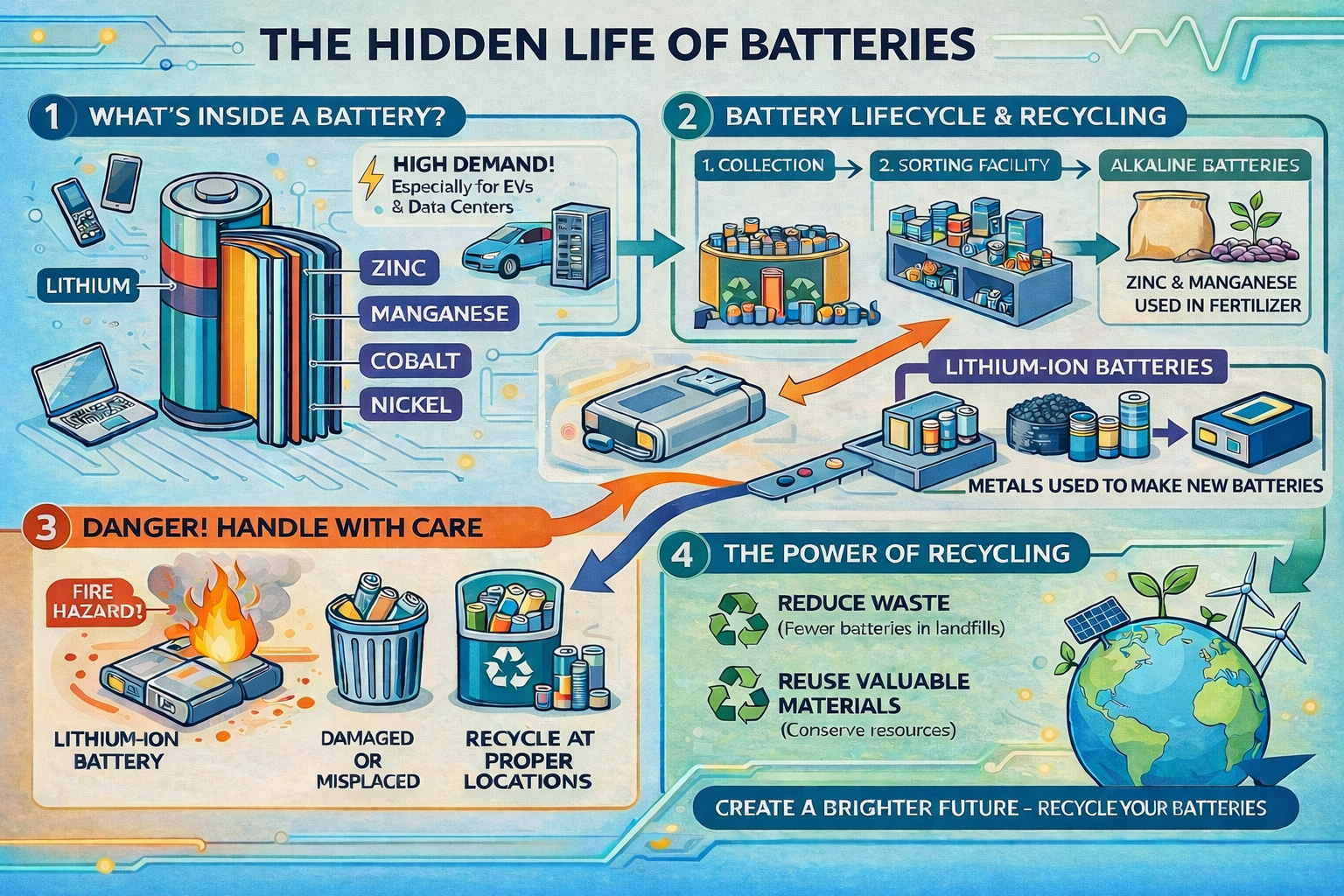
Did you know that batteries power more than your remote control or cell phone? They are used in so many small and large devices: laptops, power tools, toys, vehicles, and even data centers. Inside every battery, there are important materials like lithium, nickel, cobalt, zinc, or manganese. All of these materials store and release energy. This is what allows the devices to work. As inventors and entrepreneurs create and spread new technologies, the demand for these materials increases. Today, data centers and energy storage systems (ESS) are some of the biggest consumers of these materials.
When a battery stops working, there are still materials inside the battery that can be reused. All over the nation and even the world, there are recycling facilities that will sort and process used batteries to extract the critical materials to be used again. Alkaline batteries, like those which might be in your flashlight, can be processed into a zinc and manganese concentrate, which can be used as a source of micronutrients in fertilizers. Lithium-ion batteries are made of valuable metals such as lithium, copper, manganese, cobalt, and nickel. Once a battery reaches end-of-life, it can be collected, recycled, and processed into "black mass," which contains all the valuable metals that make up battery anodes and cathodes. The typical black color is due to the high concentrations of graphite contained in the anodes of batteries. These important materials can then be used over and over again to make new batteries.
The key is to safely and properly handle the recycling and processing of used batteries. If they are damaged or thrown into the wrong place (like the trash or curbside recycling bins), especially lithium-ion batteries, then they can cause thermal events (fires). This happens because the materials inside can react when exposed to the air, flammable objects, or chemicals. This is why it is so important not to simply throw your batteries into the trash, and not to put them into a regular or curbside trash or recycling bin, but instead to take them to a proper recycling location. This helps us to reduce waste and maintain a safer environment.
Give students time to read and process the content. Then introduce the next section, using the See-Think-Wonder thinking routine.
Watch the video and complete the routine: Web Link - How Millions Of Batteries Are Recycled Every Day | YouTube
| SEE What do you notice? |
THINK How does this facility help us understand safe recycling? |
WONDER What solutions might improve this system? |
Now that you've read about batteries and watched a video of a battery recycling center, it's time to pause and check your understanding.
Check Your Understanding
- What is a battery's main job?
- What does it mean when we say a battery stores and releases energy?
Batteries store chemical energy. When connected to a circuit, chemical reactions occur, releasing electrical energy. This energy powers the device, while the battery gradually depletes its stored chemicals. The battery converts chemical energy into electrical energy that makes devices work.
Figure 1: The Hidden Life of Batteries, image created from JASON Learning prompts by ChatGPT, March 2026
Battery Types Investigation
Explore Web Link - Battery Types | Cirba Solutions to learn about battery types.
Use Supporting Material - Battery Types - Graphic Organizer to record your learning and discoveries as you explore and ask questions. Choose two batteries from each section of the Cirba webpage to record your discoveries.
| Household Batteries Investigation Battery One | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
| Household Batteries Investigation Battery Two | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
| Industrial Batteries Investigation Battery One | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
| Industrial Batteries Investigation Battery Two | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
| Vehicle Batteries Investigation Battery One | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
| Vehicle Batteries Investigation Battery Two | |||
|---|---|---|---|
| Where is this battery commonly used? | Rechargeable or Single-use? | What are the advantages? | What are the challenges? |
Check Your Understanding
- Which battery format will last the longest?
- Which battery would be best for everyday household use?
- When using lithium-ion batteries, how can you reduce risks?
- Which battery chemistry has a larger environmental impact? Why?
In your journal (digital or paper), answer: Why is it important to choose the best battery for the device being used?
Mini-Lab Observe and Classify
Your teacher will pass out a variety of batteries. With your group, observe and classify the batteries using the following criteria:
- Size
- Labels
- Uses
- Possible materials
For example, these are Duracell AA batteries and can be used in toys, remotes, etc. Be sure to justify your thinking in your group discussion.
Connect-Extend-Challenge
Watch this video from Rumpke Waste and Recycling: Web Link - The Ways and Whys of Recycling with Jeff Snyder of Rumpke Waste & Recycling
Use the thinking routine Connect-Extend-Challenge from Project Zero to help you process and think deeply about the ideas presented in the video.
| Connect How is this like other recycling? |
Extend What surprised you? |
Challenge Why do people still use trash bins? |
Share What You Learned
Choose how you will share what you have learned. Make sure to get your instructor's approval before sharing your creation with others.
Choice One:
Create a flowchart, either by hand or digitally, showing how two different types of batteries are recycled and used to produce a new product. Be sure to include labels, flow shapes to show the system process, and visual elements to communicate the process.
Choice Two:
Create a poster or digital slide (Google, PowerPoint, Canva, etc.) that creates awareness for your audience about the importance of recycling batteries safely and properly to reduce risks, especially for lithium-ion batteries.
Choice Three:
Research and share with others where local battery recycling is taking place in your community. Create a flyer about the importance of recycling batteries safely and where they can be properly recycled.